Global Microbial Threats in the 1990s

At the present time there is no Government agency or interagency group that has the mandate, the flexibility, or the funds necessary to respond to international infectious disease emergencies. Therefore, the capacity of the U.S. Government to provide assistance to control epidemics overseas is limited, at best. Concerned individuals in different Government agencies scramble to find resources and solutions on an ad hoc basis. Response is made even more difficult by the occasional occurrence of widespread shortages of drugs, vaccines, and antisera. Moreover, there is virtually no surge capacity for producing many of the unique medical supplies needed on an emergency basis.
Nevertheless, the international public health community relies heavily on U.S. expertise and on U.S.-supported institutions overseas. CDC, NIH, DoD, and USAID may provide technical assistance to WHO when a disease outbreak occurs, and USAID often provides communications facilities as well as financial and logistical support. However, CDC, DoD, and USAID (as well as other U.S. agencies) all face legal, financial, or jurisdictional obstacles when they strive to respond to international infectious disease emergencies. Our ability to respond ensures that we learn of outbreaks quickly. Thus, it is critical to retain that capability.
Medical supplies that are frequently required during an epidemic include the following.
Diagnostics
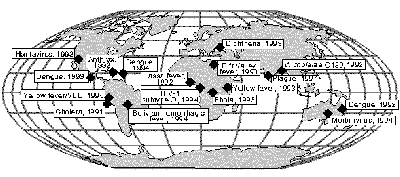
If a country suspects an outbreak of a re-emerging disease (such
as Venezuelan equine encephalitis in Peru, plague in India, or
cholera in Brazil), health practitioners may lack the diagnostic
reagents to confirm the nature of the threat. Without timely
confirmation, it is not possible to institute effective control
measures or to rule out known diagnoses if a new, or re-emerging
disease is suspected. In many cases, diagnostic reagents are
available as research tools, but not as standardized commercial
products. In such cases, training may be needed to use these
research tools appropriately.
Vaccines
If a disease is reliably diagnosed as one that is preventable by
vaccine (e.g., diphtheria and yellow fever), vaccinating people
in the surrounding area represents the most cost-effective way to
protect those people and prevent the disease from spreading.
Indeed, smallpox has been eradicated worldwide using a vaccine,
saving millions of dollars (see "Savings Due to Vaccination,"). By the year 2000, polio is targeted for eradication as
well. Logistical considerations may prevent delivery of doses of
a vaccine in time to stop transmission promptly. Where a vaccine
is appropriate, a coordinated response network can help gain
rapid access to an adequate supply of quality products that meet
local requirements. However, if supply needs are greater than can
be met by redistribution, the time required for additional
production can be lengthy. In such cases, a coordinating
infrastructure may be able to mobilize the industrial surge
capacity required to meet increased production needs.
Immunoglobulins and Antisera
Individuals who recover from certain infections produce
protective antibodies that are present in the bloodstream. When
commercially prepared serum from the plasma of former patients is
transferred by injection to another person, these antibodies (or
immunoglobulins) can provide temporary protection from that
disease when a vaccine is not available. Antisera can be made by
immunizing animals, such as horses, and the resulting
high-titered animal antisera can be used for treatment.
Immunoglobulins and antisera can be very important in some
situations, but are of limited use for long-term control. In
addition, available supplies are generally limited.
Drugs
Many infectious diseases are caused by microorganisms that are
susceptible to antibiotic drugs. However, there are occasional
regional shortages of antibiotics and in some cases, there may be
only a single manufacturer of the material from which the
finished product is made. These shortages may worsen as more
microbes become drug-resistant and fewer antibiotics remain
effective. Antiviral drugs may be of value in some situations,
and appropriate studies may be needed to determine their
effectiveness.
When an epidemic occurs, diagnostics, drugs, vaccines, and/or antisera are often needed in considerable quantities. However, in many cases they may not be available in sufficient quantities or at the necessary site, and there may be no company or agency with the surge capacity to produce more of them. The supply of medical products is driven by multiple factors, including international market forces; the needs anticipated by U.S. agencies, foreign governments, and international organizations; and the needs stimulated by Government incentives, such as the Orphan Drug Act and national vaccine plans. Some supplies may be available from other countries. However, most countries have national quality control requirements that need to be met. Harmonization of standards internationally along with strengthened local enforcement may enhance the acceptance of emergency medical supplies.
When a drug or vaccine shortage is extensive, the best solution may be to initiate production of additional supplies. However, emergency production is often hampered by insufficient manufacturing facilities, the complexity of production methods, the length of time needed for production and quality control testing, licensure requirements, concerns over liability issues, distribution problems, and/or lack of funds. These difficulties can cause significant delays, giving diseases time to spread. Early detection and rapid coordination should shorten the response time.
Moreover, if the disease is newly emergent, and no treatments or vaccines are available, it is necessary to mobilize the research and public health communities to begin seeking new solutions.
In times of need, interagency Government groups and ad hoc committees try to find remedies as best they can. FDA, CDC, and pharmaceutical manufacturers have successfully worked to redistribute available supplies of drugs, vaccines, immunoglobulins, and antisera, and to accelerate the production, testing, and release of new supplies to meet shortfalls. In certain emergencies they have resorted to modifying immunization schedules. When necessary, ad hoc coordination of activities of the NIH, CDC, FDA, DoD, and USAID has redirected resources to research and development efforts. In the vaccine area, coordination activities have been assigned by legislation to the Director of the National Vaccine Program Office. The FDA has limited authority under the Public Health Service Act to prepare biologic products for FDA or other agencies' use, although resources are required to utilize this authority. Both the National Vaccine Plan and the draft Pandemic Influenza Plan contain provisions that authorize payment for some production of vaccines against influenza and childhood diseases, for use in the United States. However, in most cases there is no U.S. Government mechanism to oversee procurement or production of emergency medical supplies.
There is a critical need for coordination and strategic planning to rethink and upgrade efforts for emergency preparedness for responding to disease outbreaks. Many groups are needed to participate in this effort, including CDC, NIH, and FDA; the DoD, the Department of State, and USAID; state and local health departments; and the private sector, including pharmaceutical trade organizations and research universities.
To fulfill this function, a designated U.S. Government group
might identify potential suppliers of the drug or vaccine and
enter into production agreements with one or more of them who
might require
To aid in this work, it is recommended that the Task Force
 Return to CISET Report Home Page
Return to CISET Report Home Page