Global Microbial Threats in the 1990s

As it turned out, our collective -- and quite understandable -- euphoria did not take into account the extraordinary resilience of microbes, which have a remarkable ability to evolve, adapt, and develop resistance to drugs in an unpredictable and dynamic fashion. Moreover, disease-carrying insects have developed resistance to pesticides in a very short time.
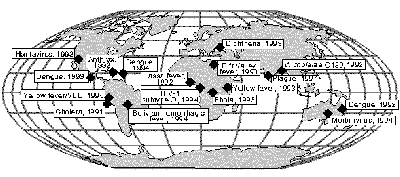
Today, most health professionals agree that new microbial threats are appearing in significant numbers, while well-known illnesses thought to be under control are re-emerging. Most Americans are aware of the epidemic of the acquired immunodeficiency syndrome (AIDS) and the related increase in tuberculosis (TB) cases in the United States. In fact, there has been a general resurgence of infectious diseases throughout the world, including significant outbreaks of cholera, malaria, yellow fever, dengue, and diphtheria, as well as illnesses caused by antibiotic-resistant bacteria. There has also been a resurgence of fungal infections for which there are very few treatments. Furthermore, the incidence of AIDS is increasing in many countries.
New diseases have also appeared within the United States, including Lyme disease, Legionnaires' disease, and most recently, hantavirus pulmonary syndrome (HPS). HPS was first recognized in the southwestern United States in 1993 and has since been detected in more than 20 states and in several other countries in the Americas. Other new or re-emerging threats in the United States include multi-drug resistant TB, antibiotic-resistant staphylococcal, enterococcal, and pneumococcal infections, and diarrheal diseases caused by the parasite Cryptosporidium parvum and by certain strains of Escherichia coli bacteria. In fact, only one antibiotic remains consistently effective against common hospital-acquired staphylococcal infections. Meanwhile, the number of new antibiotics introduced into the U.S. market has declined; not one new antibiotic was approved in 1994. In the race between drug-resistant bacteria and new drugs, the resilient bacteria are winning.
Smallpox. The economic benefits of the Smallpox Eradication Program have been substantial for all of the countries in the world, as preventive measures and treatment facilities for smallpox are no longer needed. The cost to the United States for the successful 13-year campaign to eradicate smallpox throughout the world was about $30 million. Since smallpox was eradicated in 1977, the total investment has been returned to the United States every 26 days.
Polio. Once a common cause of disabilities or death, polio has been eliminated from the Americas. The current drive towards global eradication is one of the great challenges of our generation. Once the global eradication program is completed (t arget date: 2000), the United States will save millions of dollars yearly on vaccination costs alone, since there will no longer be a need to immunize newborns. Based on the current rate of progress toward eradication, WHO predicts a global savings of $5 00 million by the year 2000, increasing to savings of $3 billion annually by the year 2015.
Other Infectious Diseases of Childhood. Health economists estimate that for every dollar spent on the measles/mumps/rubella vaccine, $21 are saved; for the diphtheria/tetanus/pertussis vaccine, $29 are saved; and for the polio vaccine $6 are saved.
Without preventive public health measures in the United States and abroad, uncontrolled outbreaks can grow into major epidemics. However, our earlier successes in controlling infections have bred complacency, and the components of the U.S. public health system that protects the public from infectious microbes have been neglected, concentrating their resources on a few targeted diseases.
Nevertheless, the subject of emerging infectious diseases is beginning to receive sustained public attention. In 1992, the Institute of Medicine's report, "Emerging Infections: Microbial Threats to Health in the United States," clarified the issue of emerging diseases for policymakers in government and in academia. In response, the CDC issued the 1994 report "Addressing Emerging Infectious Disease Threats: A Prevention Strategy for the United States." Other U.S. Government agencies, including NIH, USAID, and DoD have also examined the issue of U.S. vulnerability to epidemics and re-emerging health problems.
Quite recently, public discussion has been further focused on the global issue of emerging diseases by the publication of two best-selling, non-fiction books, The Hot Zone by Richard Preston, and The Coming Plague by Laurie Garrett, and by popular movies such as "Outbreak," starring Dustin Hoffman. Concerns about antibiotic-resistant bacteria and food-borne diseases, as well as the recent plague outbreak in India and the Ebola outbreak in Zaire, have been widely discussed in many news magazines, in print and on television. This media attention has informed the American public of the reasons why it is in our national interest to strengthen disease surveillance and control efforts internationally.
Thus, the effort to build a global surveillance and response system is in accord with the national security and foreign policy goals of the United States. Moreover, leadership in global infectious disease surveillance and control is a natural role for the United States. American business leaders and scientists are in the forefront of the computer communications and biomedical research communities (both public and private sector) that provide the technical and scientific underpinning for disease surveillance. Furthermore, American scientists and public health professionals have been among the most important contributors to the international efforts to eradicate smallpox and polio.
The challenge ahead outstrips the means available to any one country or to international organizations. The U.S. Government must not only improve its capacity to meet the growing threat of emerging infectious diseases, but also work in concert with other nations and international bodies. Although international efforts must be coordinated to prevent global pandemics, disease surveillance must be the responsibility of each sovereign nation. However, individual governments may not easily share national disease surveillance information, fearing losses in trade, tourism, and national prestige. Nevertheless, because U.S. experts are often consulted on problems of infectious disease recognition and control, the U.S. Government is usually informed about major disease outbreaks in other countries, although not always in an official or timely fashion. To ensure that we continue to be notified when an unusual outbreak occurs, we must encourage and support other countries' efforts in national disease surveillance and respond when asked for assistance. We must strive to develop a sense of shared responsibility and mutual confidence in the international effort to combat infectious diseases.
There is much room for optimism. If the United States takes the lead, we can expect that other nations will contribute resources to a global surveillance system. Both Canada and the European Union have recently decided -- in spite of tight budgets -- to provide substantial funds ($7 and $10 million per year, respectively) to strengthen infectious disease surveillance and control. It is also absolutely critical that developing nations be engaged in an international effort that is in their own interests. In May 1995, WHO passed a resolution urging member states "to strengthen national and local programmes of surveillance for infectious diseases, ensuring that outbreaks of new, emerging, and re-emerging infectious diseases are identified." Soon after the resolution was drafted, WHO issued a report urging the strengthening of global disease surveillance and control, and encouraging greater use of WHO Collaborating Centers in this endeavor.
The direct and indirect costs of infectious disease are staggering (see Table 1). Clearly, public health measures that prevent infectious diseases can be extremely cost-effective. In 1994 and 1995 two major U.S. health-care expenses have been for the treatment of tuberculosis and AIDS. The Public Health Service budget for fiscal year 1996 includes $343 million to combat TB and nearly $3 billion to combat AIDS. TB is a very old, well-known disease that has re-emerged sometimes in a drug-resistant or multidrug-resistant form. AIDS, on the other hand, is a new disease, unrecognized before the 1980s. When the first cases of AIDS and drug-resistant TB were detected in the United States, control measures were delayed, partly because of a lack of surveillance information and incomplete understanding of the epidemiology of these diseases.
Table 1 Estimated costs of common infectious diseases in the United States
Tuberculosis
For many years, the United States had in place a surveillance
system to monitor cases of TB. However, during the 1980s, federal
and local spending on infectious disease control declined, and in
1986 the surveillance system for multidrug-resistant TB was
discontinued. Consequently, there was no warning signal when
drug-resistant TB emerged in the late 1980s. This lack of early
warning undoubtedly contributed to the more than $700 million in
direct costs for TB treatment incurred in 1991 alone.
Surveillance of drug-resistant TB was not reinstated until 1993,
by which time multidrug-resistant TB had became a public health
crisis and millions of federal dollars had been appropriated.
AIDS
As mentioned above, AIDS is a new disease that was unknown before
the 1980s, and thus, was not on any surveillance lists. AIDS
weakens the immune system, allowing other infections to take
hold. Therefore, it can be difficult to diagnose since its
clinical presentation may involve a variety of symptoms, and its
incubation period (the time between infection and the appearance
of symptoms) can be many years. Nevertheless, long before AIDS
was diagnosed in the United States and Europe, a distinct
syndrome called slim disease (now known to be a form of AIDS)
that causes its victims to waste away was recognized by African
doctors. In fact, an aggressive, slim-associated, generalized
form of Kaposi sarcoma, distinct from the classical form, has
been described in Uganda since at least 1962. Some health workers
believe that if a global surveillance network had been in place
in the 1970s, AIDS might have been identified earlier, perhaps
before it became well established in the United States.
Epidemiologists might have gained a head start in learning how
AIDS is transmitted and prevented, and many lives might have been
saved. However, other health experts believe that the lack of
disease surveillance and specimen collection facilities in
central Africa in the 1960s and 1970s make it nearly impossible
to be sure, even in retrospect, if AIDS was present at that time.
Table 2 Examples of pathogenic microbes and infectious diseases
recognized since 1973
Table 3 Re-emerging infections during the last two decades and
factors contributing to their re-emergence
In recent years, antimicrobial drug resistance has become a
serious problem in the United States and abroad. Antimicrobial
resistance occurs when a disease-carrying microbe (bacteria,
virus, parasite, or fungus) is no longer affected by a drug that
previously was able to kill the microbe or prevent it from
growing.
The types of antimicrobial drug resistance include
Antibiotic resistance. Resistance to drugs that kill bacteria or
keep them from growing. Antibiotic resistance is a growing
problem in American hospitals. It affects the treatment of
bacterial pneumonia, TB, ear infections, and many other common
bacterial illnesses.
Antiviral resistance. Resistance to drugs that prevent the
replication of viruses. Antiviral resistance is a serious problem
in the treatment of AIDS, which is caused by the HIV retrovirus.
For instance, most strains of HIV become resistant over time to
the drug AZT, which is a first-line drug against AIDS.
Antiparasite resistance. Resistance to drugs that kill parasites
or keep them from growing. For example, common medicines and
prophylactic treatments for malaria, including chloroquine, are
no longer reliably effective because drug resistance is spreading
among malarial parasites.
Antifungal resistance. Resistance to drugs that kill fungi or
keep them from growing. Drug resistance has developed to the
drugs for the treatment of candida infections which are common in
AIDS patients worldwide.
Multidrug resistance. A bacterium, parasite, or fungus which has
developed resistance to several previously potent drugs,
sometimes through a non-specific mechanism that allows the
microbe to eject or neutralize drugs of different chemical
structures. In the United States, multidrug-resistant TB is on
the rise.
Pesticide resistance. A microbe-carrying insect or animal
(disease vector) becomes resistant to an agent that previously
was used to kill it. The most common type of pesticide resistance
is insecticide resistance. Insecticides are used in many parts of
the world to kill mosquitoes that carry malaria parasites. Other
insect vectors include tsetse flies (which carry parasites that
cause African sleeping sickness) and reduviid bugs (which carry
parasites that cause Chagas' disease, a serious disease prevalent
in South America.)
Researchers at CDC's biosafety level-four laboratory in Atlanta,
Georgia, confirmed on May 10 that a mysterious disease outbreak
in Kikwit, Zaire, was caused by the deadly Ebola virus. On the
following day, the Government of Zaire informed its citizens of
the danger and began to institute quarantine measures. At the
government's invitation, WHO investigators arrived in the capital
city, Kinshasa, on May 10, where a 3-person CDC team joined them
on May 11.
A few days earlier, on May 6, the U.S. Embassy in Zaire had
learned that Kikwit, an area about 350 miles from Kinshasa, was
suffering an outbreak of an unusual hemorrhagic fever.
A medical professor at the University of Kinshasa reported that
the symptoms of the fever patients were the same as those seen in
an earlier Ebola outbreak (in 1976). The Ebola virus, which is
transmitted through contact with infected bodily fluids, causes a
fatal illness in 50-90 percent of its victims, and there is no
known drug treatment or vaccine.
The Government of Zaire has quarantined the Kikwit area and
closed the road leading from Kinshasa to Bandundu State, where
Kikwit is located. The U.S. Embassy has declared the outbreak a
disaster, and USAID's Office of Foreign Disaster Assistance
(OFDA) has authorized the payment of $25,000 to non-governmental
organizations (NGOs) in the area for the purchase and transport
of disposable protective clothing, plasma, body bags, and
essential medicines and supplies. OFDA has also requested a
Department of Defense airlift to transport equipment and
supplies, including plasma, plastic hospital gowns and sterile
needles.
The Vice Prime Minister of Zaire, Kamanda Va Kamanda, accompanied
the WHO and CDC doctors to Kikwit on May 12, where the
international team set about its primary task of containing the
outbreak of Ebola fever. As part of that effort the team is
trying to trace the outbreak's first casualty to gain clues to
the virus's animal or insect host (its "reservoir"). The
international team has been joined by additional doctors from
Zaire and elsewhere, including government and NGO medical workers
from Belgium, South Africa, and Sweden.
The different national groups that make up the WHO-led
international team bring different resources and types of
expertise to the cooperative effort. For instance, Belgian
doctors from the organization Medicins Sans Frontieres focus on
providing clinical care and specialize in building and operating
safe, sanitary, functional hospitals and clinics. Zairian doctors
from the University of Kinshasa are familiar with most local
health problems and take the lead in clinical diagnosis, case
management, and clinical work-up. The CDC team provides expertise
in filoviruses (the class of virus to which Ebola belongs),
experience in disease surveillance and case investigation, and
access to laboratory diagnosis via the facilities in Atlanta.
Lessons from Kikwit. It is useful to examine the international
team's experiences in Zaire for ideas on how to improve U.S.
preparedness for controlling infectious diseases outbreaks in
countries with poorly developed health and communications
infrastructures. One week into the investigation, the three CDC
investigators report that the team's efforts are hampered by
difficulties with transportation and communication, and by lack
of money and personnel. Because the average incubation time (the
time between infection and the appearance of symptoms) for Ebola
is 7 days, each week's delay in instituting control measures
means that a new generation of the virus has time to spread.
1) Transportation. To investigate suspected Ebola cases, doctors
must be able to travel quickly from community to community in an
area where there are few paved roads and no public
transportation. The USAID mission to Zaire, which in past years
could be relied on for assistance with logistics and
organization, was closed in 1994. The U.S. Embassy and OFDA have
provided some help, as the CDC team did not arrive in Zaire with
authorization to purchase or rent cars or bicycles.
2) Money. The CDC team in Kikwit has no funds at their disposal
to obtain radios, cars, bicycles, or additional medical supplies.
An initial $20,000 was spent on essential equipment and medical
supplies. A week into the investigation, the team has requested
$781,000 to allow six doctors to work in Zaire for three months.
In comparison, the team that responded to the hantavirus outbreak
in New Mexico in 1994 involved 24 people working for 18 months at
a cost of 4.5 million dollars. (Note: On May 23 OFDA allocated
$750,000 for the CDC team and USAID's Bureau of Global Programs
Field Support and Research supplied another $43,000.)
3) Personnel. The Zairian medical authorities have requested that
the CDC send three additional epidemiologists and one
operations/logistics manager to provide help with travel,
communications, and procurement. In the United States, at CDC's
biosafety level-four laboratory in Atlanta, additional
technicians are needed to process the hundreds of potentially
dangerous clinical samples sent from Zaire. The international
team (not only the CDC doctors) are dependent upon the efforts of
this unique laboratory. (Update: The funds provided by USAID/OFDA
on May 23 will be used to support additional personnel in Zaire.)
4) Communication. To prevent the spread of Ebola fever, medical
workers must report all suspicious fever cases to the national
health authorities so that appropriate follow-up measures can be
instituted. There are very few telephones and no radio station in
Kikwit, although radio transmissions are received from Kinshasa.
The lack of reliable communication has hampered the international
team members' initial efforts to coordinate with each other and
the national health authorities of Zaire.
Disease Financial Cost
These costs, combined with dollars spent on AIDS and TB, exceed $120 billion per year.
Intestinal infections $23 billion in direct medical costs and
lost productivity
Food-borne diseases $ 5-6 billion in medical and
productivity costs
Sexually-transmitted $ 5 billion in treatment costs
diseases(excluding
AIDS)
Influenza $ 5 billion (direct medical costs) and
$12 billion (lost productivity costs)
Antibiotic-resistant $ 4 billion in treatment costs and increasing
bacterial infections
Hepatitis B virus infection $720+ million in combined direct
and indirect costs
Year Microbe Type Disease
1973 Rotavirus Virus Major cause of infantile diarrhea
worldwide
1975 Parvovirus B19 Virus Aplastic crisis in chronic
hemolytic anemia
1976 Cryptosporidium parvum Parasite Acute and
chronic diarrhea
1977 Ebola virus Virus Ebola hemorrhagic fever
1977 Legionella pneumophila Bacteria Legionnaires' disease
1977 Hantaan virus Virus Hemorrhagic fever with renal
syndrome (HRFS)
1977 Campylobacter jejuni Bacteria Enteric
pathogens distributed globally
1980 Human T-lymphotropic Virus T-cell lymphoma-leukemia
virus I (HTLV-1)
1981 Toxic producing strains
of Staphylococcus
aureus Bacteria Toxic shock syndrome (tampon use)
1982 Escherichia coli
O157:H7 Bacteria Hemorrhagic colitis; hemolytic
uremic syndrome
1982 HTLV-II Virus Hairy cell leukemia
1982 Borrelia burgdorferi Bacteria Lyme disease
1983 Human immunodeficiency Virus Acquired immunodeficiency
virus (HIV) syndrome(AIDS)
1983 Helicobacter pylori Bacteria Peptic ulcer disease
1985 Enterocytozoon bieneusi Parasite Persistent diarrhea
1986 Cyclospora cayatanensis Parasite Persistent diarrhea
1988 Human herpesvirus-6 Virus Roseola subitum
(HHV-6)
1988 Hepatitis E Virus Enterically transmitted non-A,
non-B hepatitis
1989 Ehrlichia chafeensis Bacteria Human ehrlichiosis
1989 Hepatitis C Virus Parenterally transmitted non-A,
non-B liver infection
1991 Guanarito virus Virus Venezuelan hemorrhagic fever
1991 Encephalitozoon hellem Parasite Conjunctivitis, disseminated
disease
1991 New species of Babesia Parasite Atypical babesiosis
1992 Vibrio cholerae O139 Bacteria New strain associated
with epidemic cholera
1992 Bartonella henselae Bacteria Cat-scratch disease; bacillary
angiomatosis
1993 Sin nombre virus Virus Adult respiratory distress
syndrome
1993 Encephalitozoon
cuniculi Parasite Disseminated disease
1994 Sabia virus Virus Brazilian hemorrhagic fever
1995 HHV-8 Virus Associated with Kaposi sarcoma
in AIDS patients
Disease or Agent Factors in Re-emergence
Viral
Rabies Breakdown in public health measures;
changes in land use; travel
Dengue/dengue hemorrhagic fever Transportation, travel and migration;
urbanization
Yellow fever Favorable conditions for mosquito vector
Parasitic
Malaria Drug and insecticide resistance; civil
strife; lack of economic resources
Schistosomiasis Dam construction, improved irrigation,
and ecological changes favoring the
snail host
Neurocysticercosis Immigration
Acanthamebiasis Introduction of soft contact lenses
Visceral leishmaniasis War, population displacement,
immigration, habitat changes favorable
to the insect vector, an increase in
immunocompromised human hosts
Toxoplasmosis Increase in immunocompromised human hosts
Giardiasis Increased use of child-care facilities
Echinococcosis Ecological changes that affect the
habitats of the intermediate
(animal) hosts
Bacterial
Group A Streptococcus Uncertain
Trench fever Breakdown of public health measures
Plague Economic development; land use
Diphtheria Interruption of immunization program due
to political changes
Tuberculosis Human demographics and behavior;
industry and technology; international
commerce and travel; breakdown of
public health measures; microbial
adaptation
Pertussis Refusal to vaccinate in some parts of
the world because of the belief that
injections or vaccines are not safe
Salmonella Industry and technology; human
demographics and behavior; microbial
adaptation; food changes
Pneumococcus Human demographics; microbial adaptation;
international travel and commerce;
misuse and overuse of antibiotics
Cholera Travel: a new strain (O139) apparently
introduced to South America from Asia
by ship, with spread facilitated by
reduced water chlorination and also
food
Common Types of Antimicrobial Drug Resistance
Lessons Learned From the Ebola Virus Outbreak in Zaire
(Written on May 18, 1995, one week after the CDC team arrived in
Kikwit, Zaire)
Poor communication has been a problem from the beginning of the
outbreak. Although the first case of Ebola probably occurred as
early as December, 1994, the international community only learned
about the outbreak in May, after the Ebola virus had nearly 20
weeks to spread. This delay reflects the weak health care systems
and the poor state of infectious diseases surveillance in most of
Africa. Over the last ten years, with the end of the
post-colonial era, the end of the Cold War, and the decline of
Western interest in tropical medicine, the public health
infrastructures in many African countries have deteriorated.
Infectious disease surveillance is nearly non-existent, and
emerging and re-emerging diseases frequently go unreported.
 Return to CISET Report Home Page
Return to CISET Report Home Page